
Mathematical and computational approaches to cardiac arrhythmia mechanisms
Ventricular tachycardia, macroreentrant atrial tachycardia, atrial fibrillation, celiac disease research, signal analysis, substrate mapping, and translational electrophysiology.
I develop whole-heart and automaton models of myocardial electrical activation to better understand the onset and perpetuation of ventricular tachycardia, macroreentrant atrial tachycardia, and atrial fibrillation. My work links arrhythmia mechanisms to electrogram patterns, activation maps, structural substrate, and translational electrophysiology, with the aim of improving substrate mapping and ablation strategies.
I also pursue celiac disease research, with recent work focusing on disease onset, the inflammatory milieu, and quantitative frameworks for understanding progression and maintenance.
About
My research has shown how functional conduction block can form at lateral boundaries of the ventricular tachycardia isthmus, how source-sink mismatch contributes to block formation, and how fragmented or unstable electrograms can help identify arrhythmogenic substrate during therapy. More recently, I have found similar low-voltage and slow-conduction properties at atrial tachycardia bottlenecks, and have used automaton modeling to show how dispersion of refractoriness and fibrosis density can generate rotational activity in atrial fibrillation.
In parallel, I have contributed to biomedical signal analysis and informatics, and have served in major editorial roles including founding Editor-in-Chief of Informatics in Medicine Unlocked and Editor-in-Chief of Computers in Biology and Medicine.
Research themes
Ventricular tachycardia
Mechanistic modeling of post-infarct reentry, isthmus geometry, source-sink mismatch, and sinus-rhythm markers of inducible VT.
- Prediction of VT isthmus location, shape, and exit orientation
- Uniform slow conduction and low-voltage signatures during sinus rhythm
- Electrogram fragmentation and instability as markers of arrhythmogenic substrate
Atrial arrhythmias
Modeling and mapping of macroreentrant atrial tachycardia and atrial fibrillation with emphasis on bottlenecks, fibrosis, and rotational activity.
- Activation-signature prediction of macroreentrant AT bottlenecks
- Voltage-based guidance for atrial fibrillation substrate mapping
- Automaton models of refractoriness gradients and fibrillatory maintenance
Selected figures
Representative images from my work in ventricular and atrial arrhythmia mechanisms.
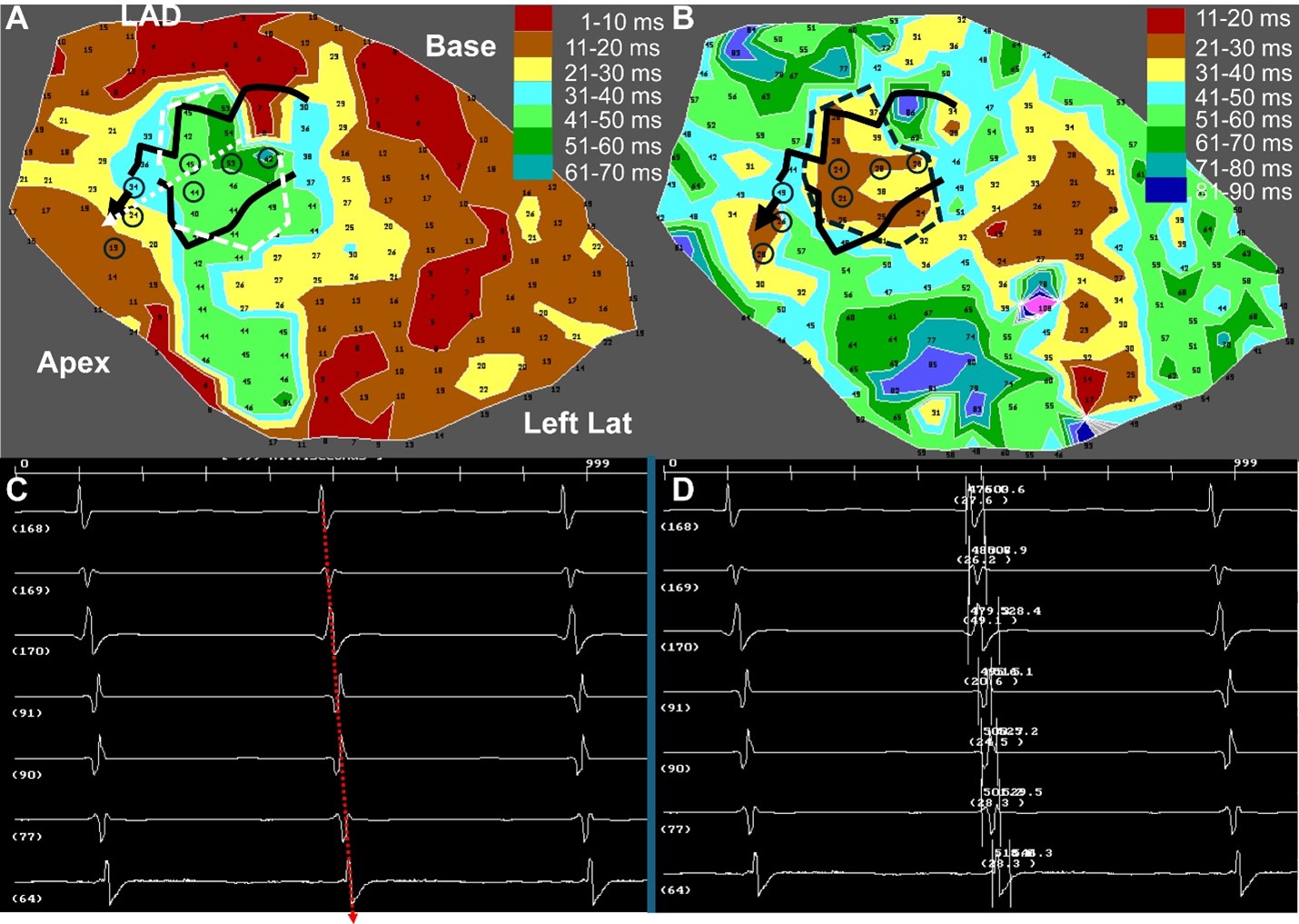
Predicting VT isthmus location from sinus rhythm maps
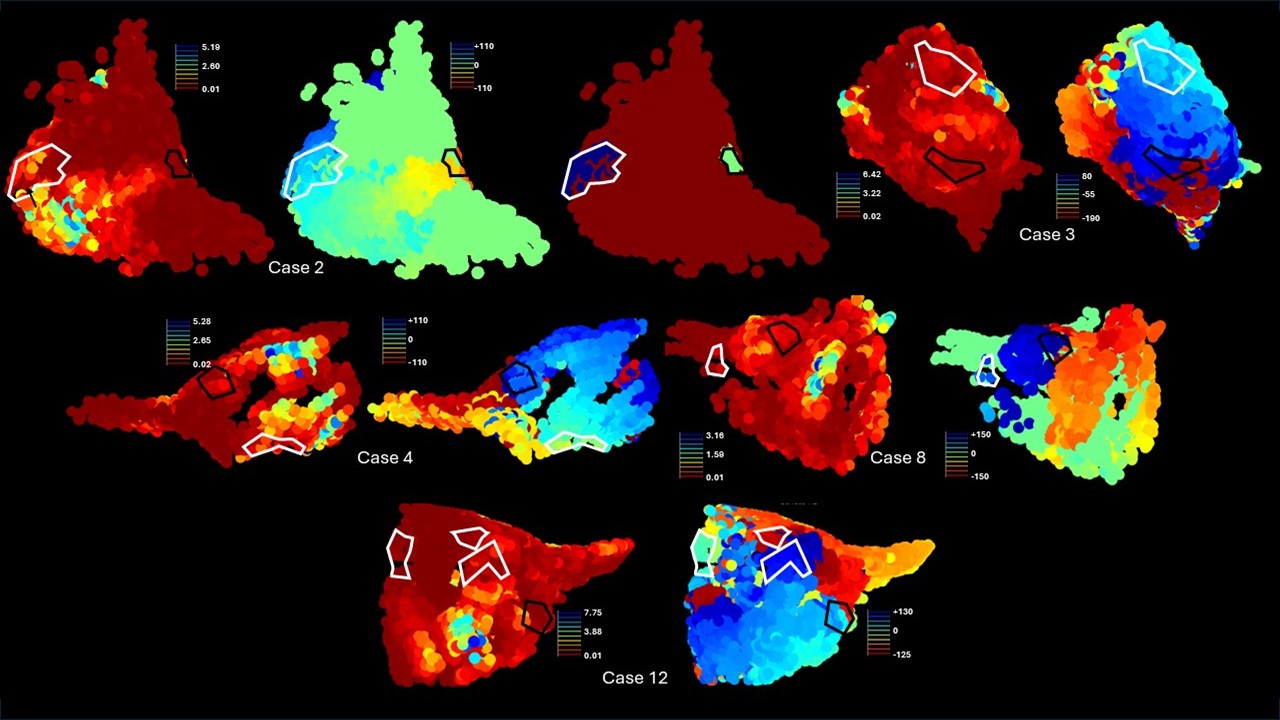
Macroreentrant atrial tachycardia targets
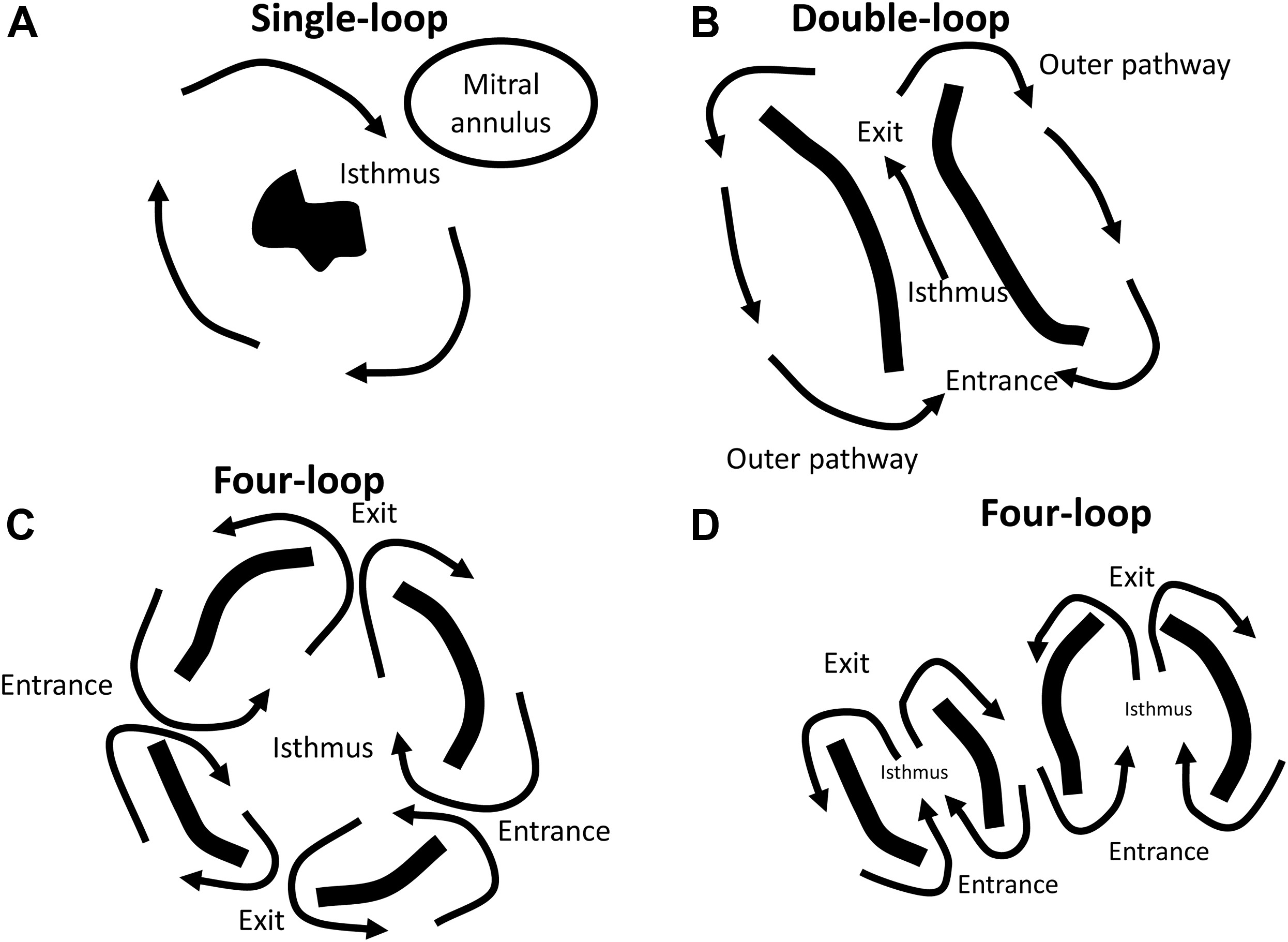
Examples of reentrant circuit patterns
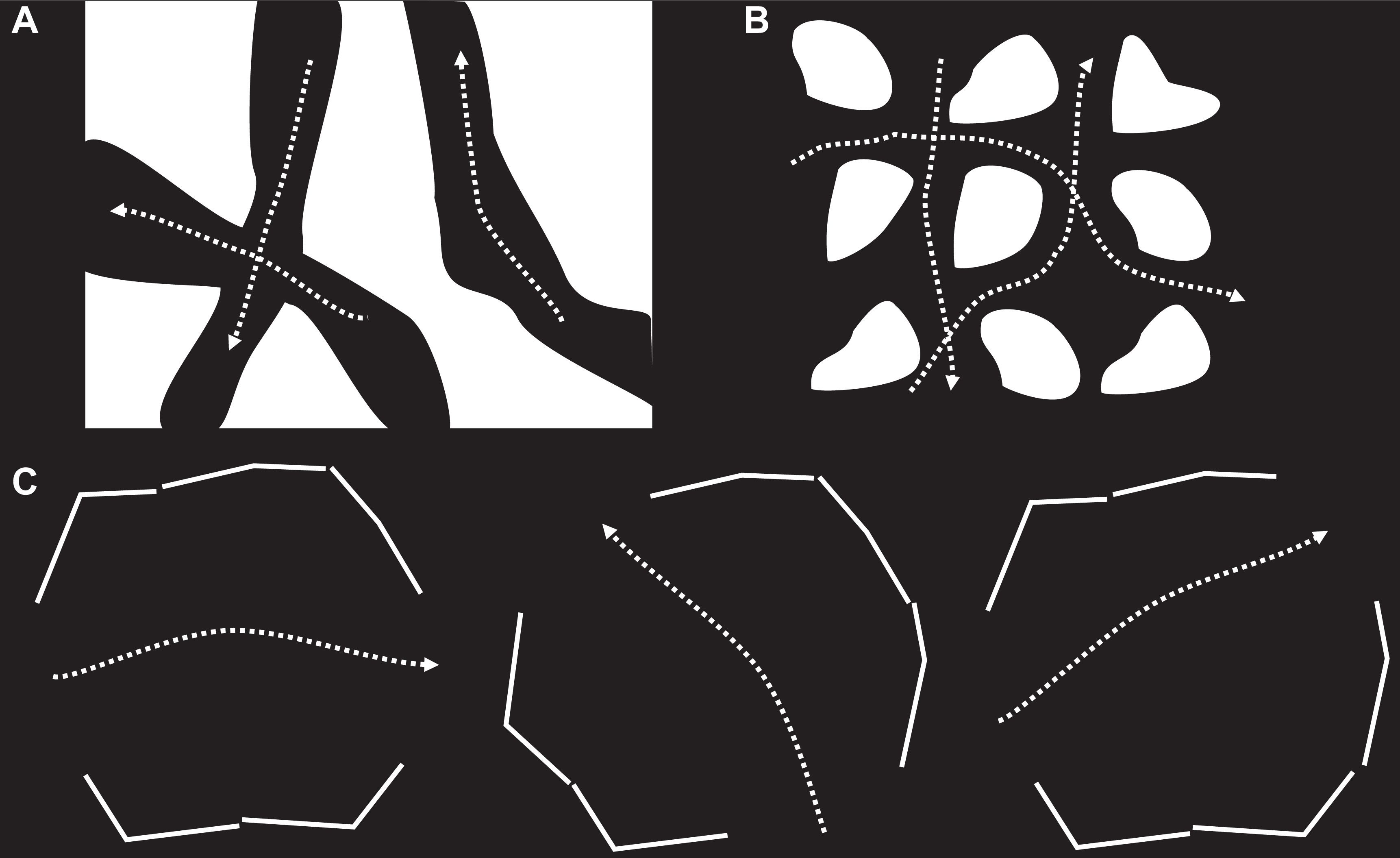
Possible conduction patterns through arrhythmogenic substrate
News
- Accepted: Celiac disease onset and the inflammatory milieu (review), World Journal of Gastrointestinal Endoscopy, 2026.
- Submitted: Onset and Maintenance of Atrial Fibrillation (review), invited to a special issue of Life, 2026.
Selected publications and roles
Recent and representative work
- Ventricular tachycardia substrate mapping: What's been done and what needs to be done. Heart Rhythm, 2025.
- Structure and function of the ventricular tachycardia isthmus. Heart Rhythm, 2022.
- Wavefront Curvature Analysis Derived from Pre-Procedural Imaging Can Identify the Critical Isthmus in Patients with Post-Infarcted Ventricular Tachycardia. Heart Rhythm, 2024.
- Activation signature valleys are predictive of macroreentrant atrial tachycardia bottlenecks where uniform low voltage and uniform slow conduction reside. Heart Rhythm, in press.
Editorial leadership and honors
- Founding Editor-in-Chief, Informatics in Medicine Unlocked (2015–2023)
- Editor-in-Chief, Computers in Biology and Medicine (2013–2021)
- Top Scholar, ScholarGPS (2024)
- Multiple invited lectureships, editorial board appointments, and international grant peer review roles
Support and collaboration
Contact
Email: ejc6@cumc.columbia.edu
Mailing address: Dr. Edward Ciaccio, HP-9-956, 180 Fort Washington Avenue, New York, NY 10032
Twitter / X: https://x.com/EdwardCiaccio
Google Scholar: Profile
NIH papers list: PubMed author query
Acknowledgment
This webpage was developed with assistance from OpenAI's ChatGPT, with final content selection, revision, and approval by Edward J. Ciaccio.